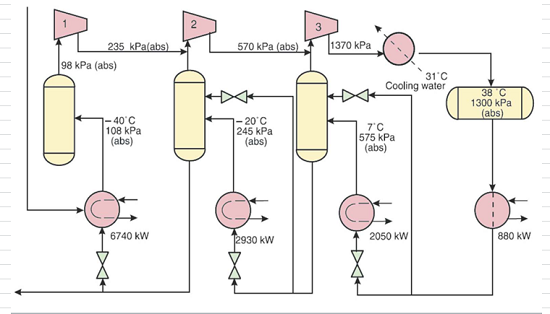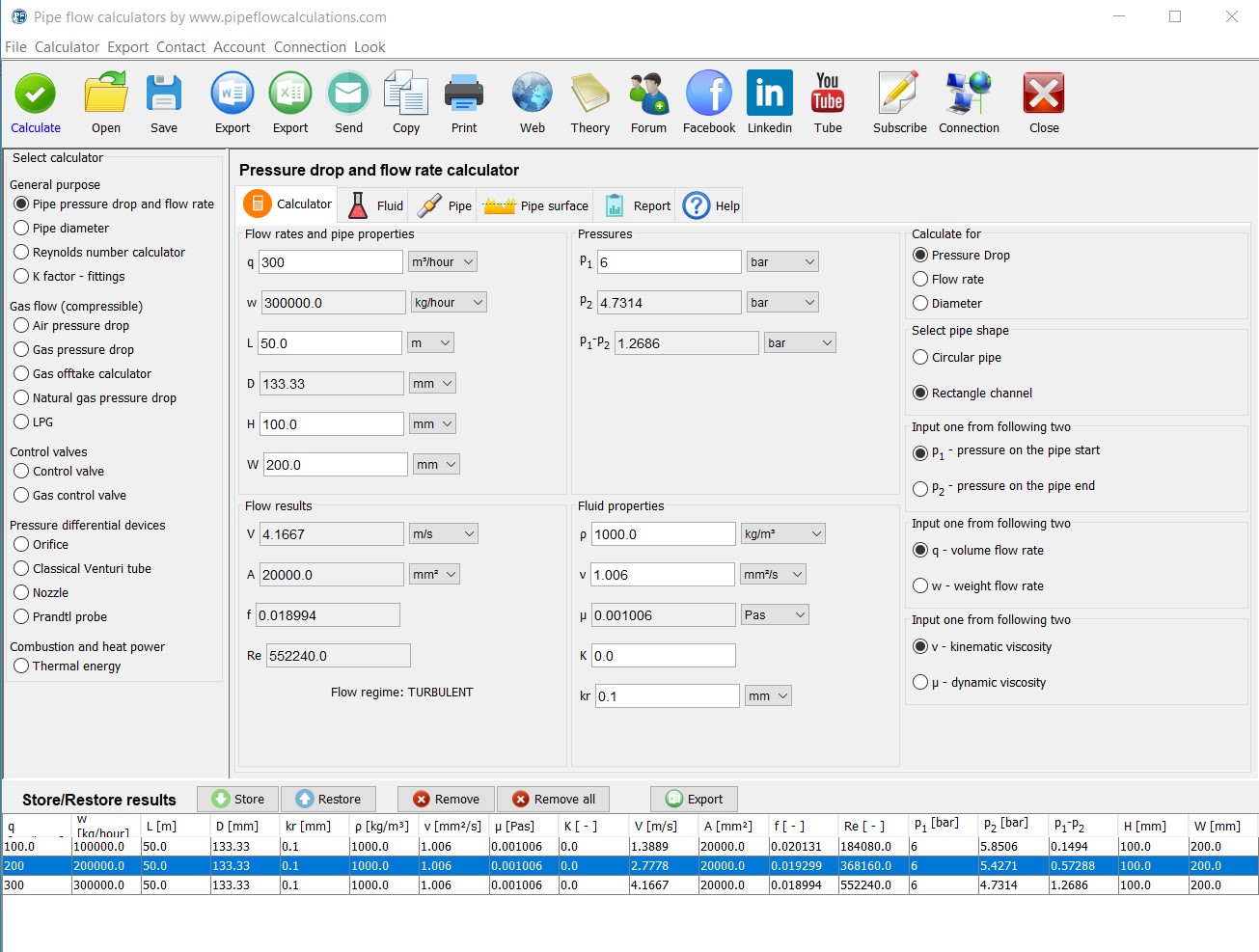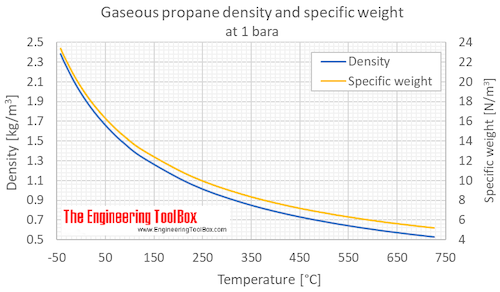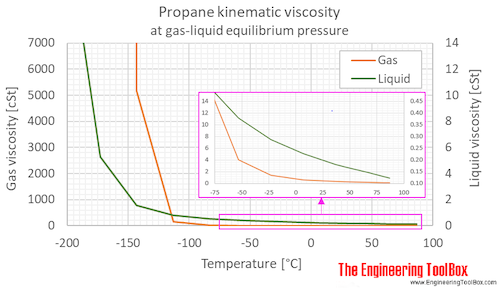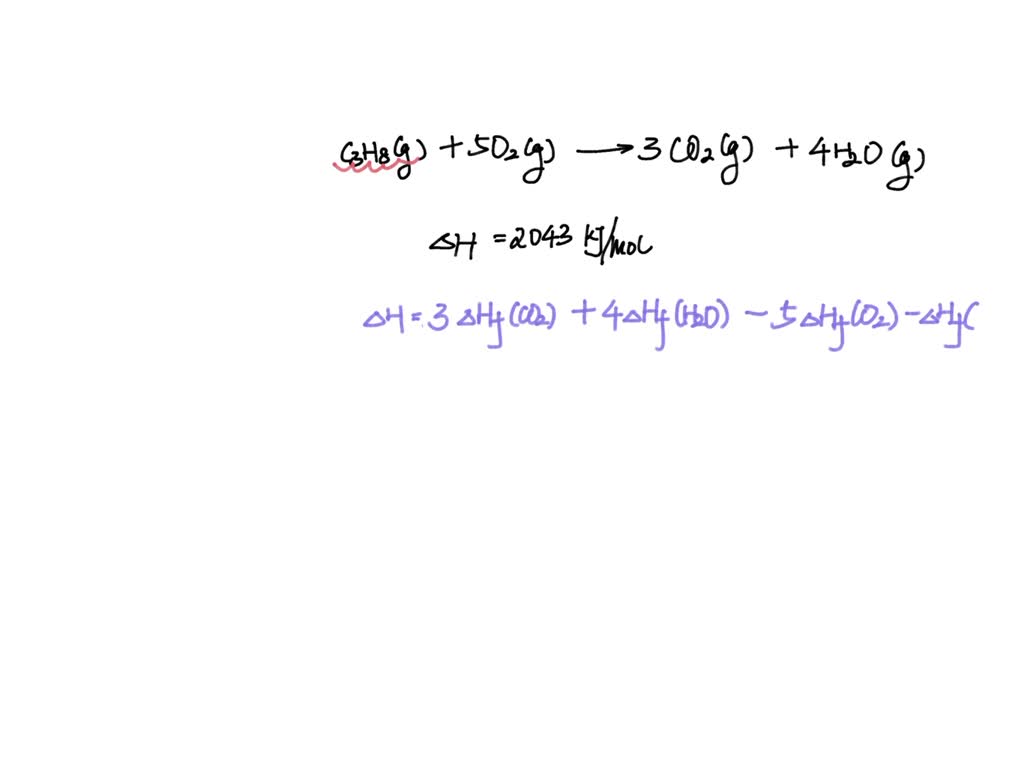
SOLVED: The standard enthalpy change for the combustion of 1 mole of propane is –2043.0 kJ. C3H8(g) + 5 O2(g) → 3 CO2(g) + 4 H2O(g) Calculate ΔfH° for propane based on
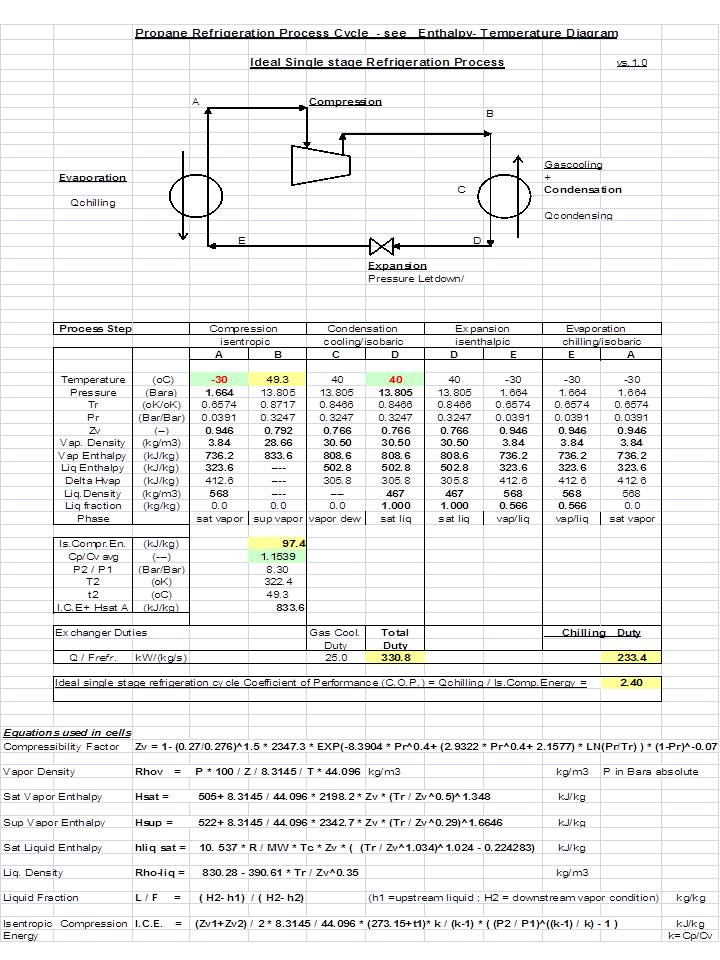
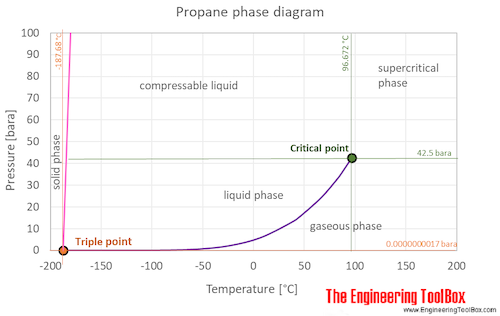
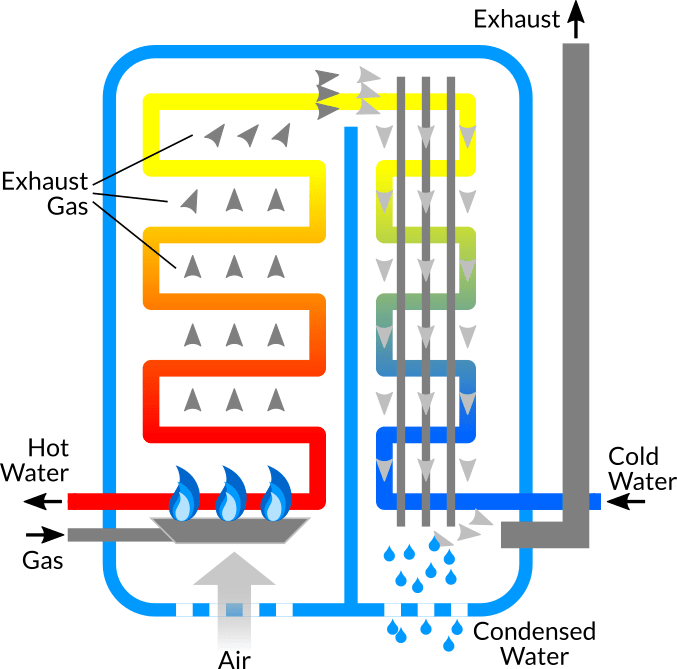
Propane Vapor and Liquid Enthalpies calculated directly with a corresponding states correlation in excel | Chem-Eng-Musings

Propane Vapor and Liquid Enthalpies calculated directly with a corresponding states correlation in excel | Chem-Eng-Musings

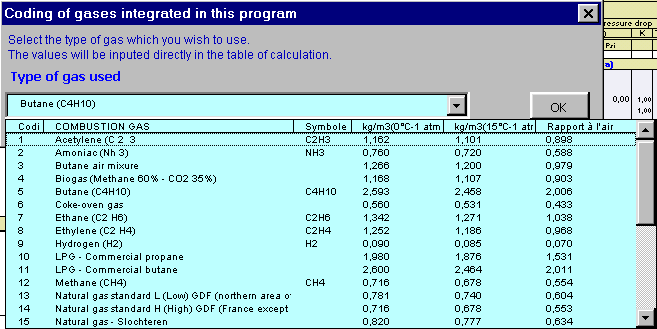
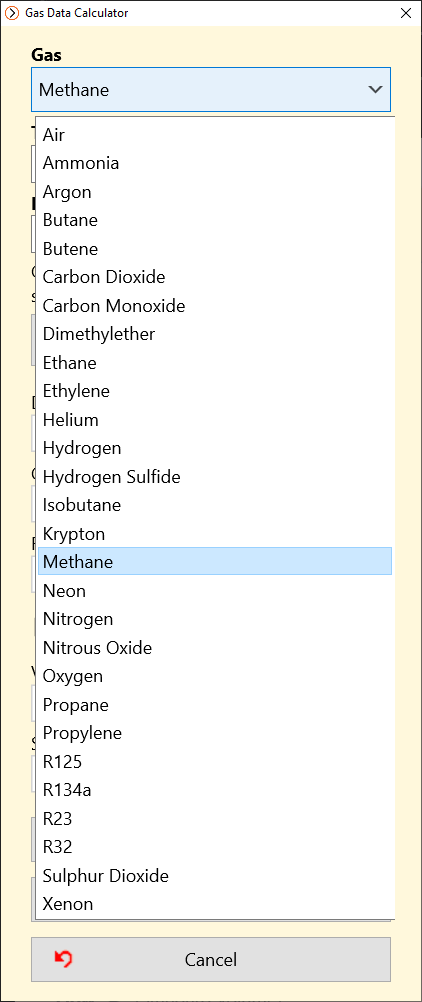
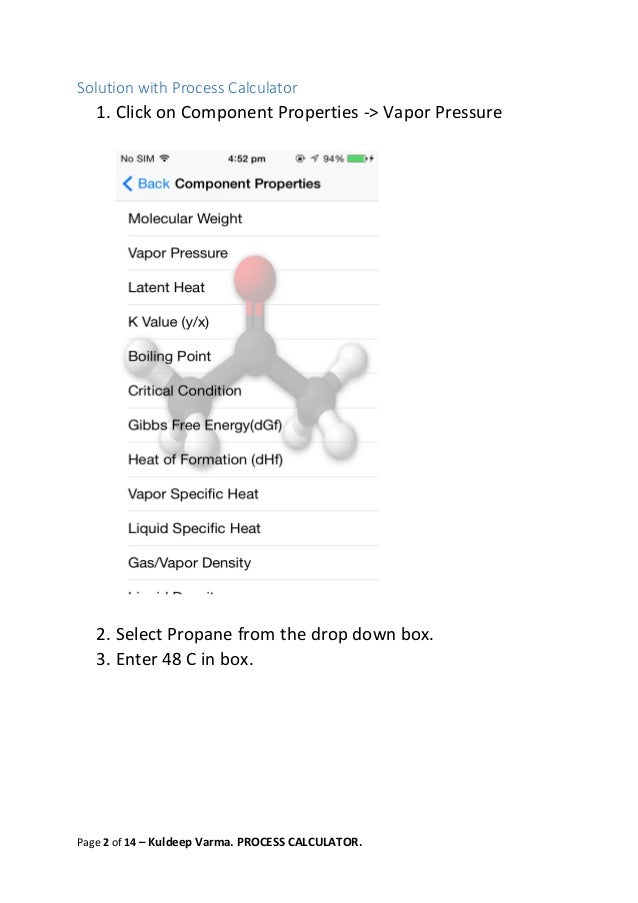
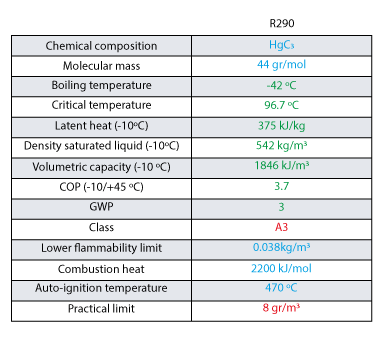
GitHub - MarkusHays/Propane-Properties: Calculate the properties of Propane using the modified BWR EOS model
GitHub - MarkusHays/Propane-Properties: Calculate the properties of Propane using the modified BWR EOS model