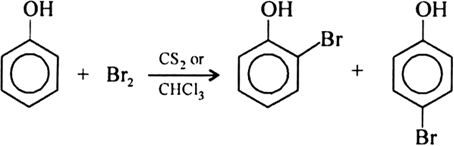
What happens when phenol reacts with bromine in solvent of low polarity like CS2or CHCl3 at low temperature? Give a mechanism for the reaction. - Zigya
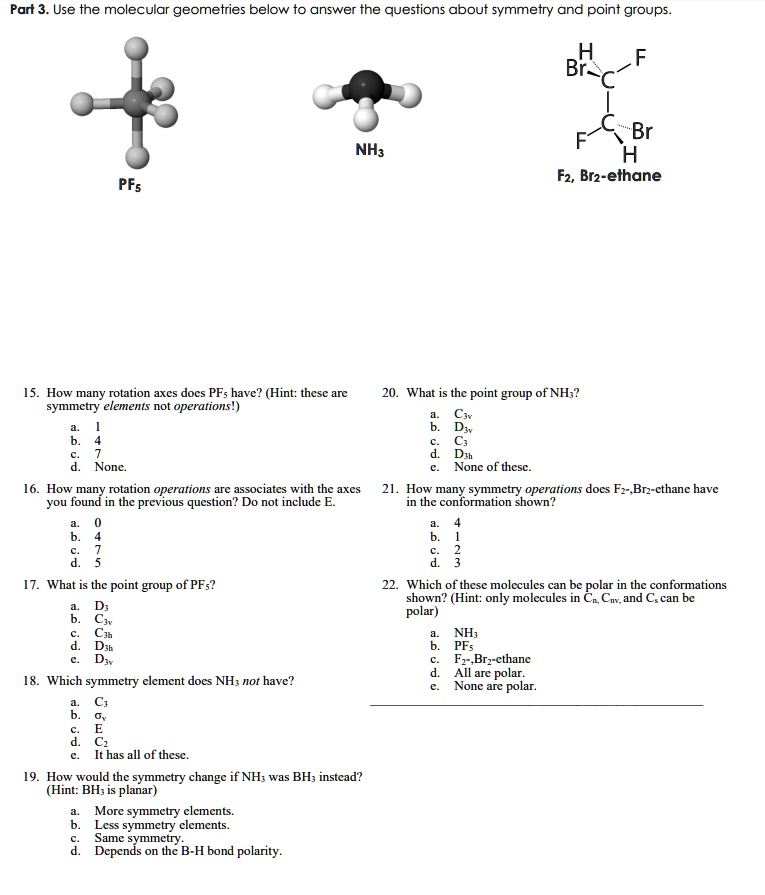
SOLVED: Part 3. Use the molecular geometries below to answer the questions about symmetry and point groups Br H F2, Br2-ethane NHs PFs How many rotation axcs docs PFs havc? (Hint: thesc

SOLVED: 1. Predict whether each compound is purely covalent, purely ionic or polar covalent: a RbCl b Sg C Ticlz d. SbCly e Lil f. Br2